Pharmaceutical Equipment Cabinet Hinges: GMP Selection Guide
Pharmaceutical equipment cabinet hinges are small components, but they can affect cleanability, cabinet sealing, corrosion resistance, access safety, and long-term maintenance in GMP-controlled environments. A hinge that works on a general industrial cabinet may create problems when exposed to frequent disinfectant wiping, controlled cleanroom airflow, stainless cabinet surfaces, and strict inspection routines.
The real issue is not simply whether the hinge can open and close the door. In pharmaceutical cabinets, poor hinge selection can create hidden crevices, exposed screw pockets, corrosion stains, particle traps, door misalignment, cleaning difficulty, and repeated maintenance work. Over time, those small hardware problems can become cleaning deviations, premature replacement, or — in audit-sensitive sites — an FDA Form 483 observation tied to equipment cleanability.
This guide is written for engineers, OEM equipment builders, cleanroom project teams, cabinet fabricators, and purchasing teams that need to specify hinges for pharmaceutical equipment cabinets. It focuses on practical selection decisions: material grade, surface finish, hinge structure, cleaning-agent exposure, door load, maintenance access, and supplier confirmation requirements.
If your project is primarily a cleanroom hardware specification — focused on ISO-classified space, tool-free access design, and stainless cabinet fabrication — the dedicated cleanroom cabinet hinge guide covers that angle. This page focuses specifically on pharmaceutical manufacturing context: GMP cleaning frequency, disinfectant chemistry, audit-relevant cleanability, and operator access during validated production runs.
Why Pharmaceutical Cabinet Hinges Need Different Selection
A pharmaceutical equipment cabinet is not the same as a general utility cabinet. It may be installed in a cleanroom, laboratory, production suite, weighing room, filling area, sampling room, or controlled storage area classified under ISO 14644-1 air cleanliness classes. In these spaces, every exposed hardware detail can affect cleaning time, residue buildup, corrosion behavior, and inspection confidence.
Common hinge-related problems include disinfectant residue trapped around hinge knuckles, rust staining around fasteners, particles released from worn pins, cabinet doors that no longer close evenly, and screw heads that create hard-to-clean recesses. These issues may not appear during first installation, but they become visible after repeated cleaning, door cycling, and maintenance access.
For pharmaceutical equipment, hinge selection should begin with the cabinet’s operating environment: cleaning method, chemical exposure, door weight, access frequency, room classification, and maintenance procedure. The hinge should support the cleaning strategy, not become the part that makes cleaning harder.
Failure Chain: From Hinge Crevices to Cleaning Deviation
The problem chain usually starts small. A surface-mounted hinge with exposed gaps may collect moisture or residue. If the fastener, hinge pin, or hinge leaf is not compatible with the cleaning agent, corrosion or staining can begin. As the hinge wears, door alignment may change. Once alignment changes, the cabinet door may not seal or close consistently, increasing cleaning time and inspection uncertainty.
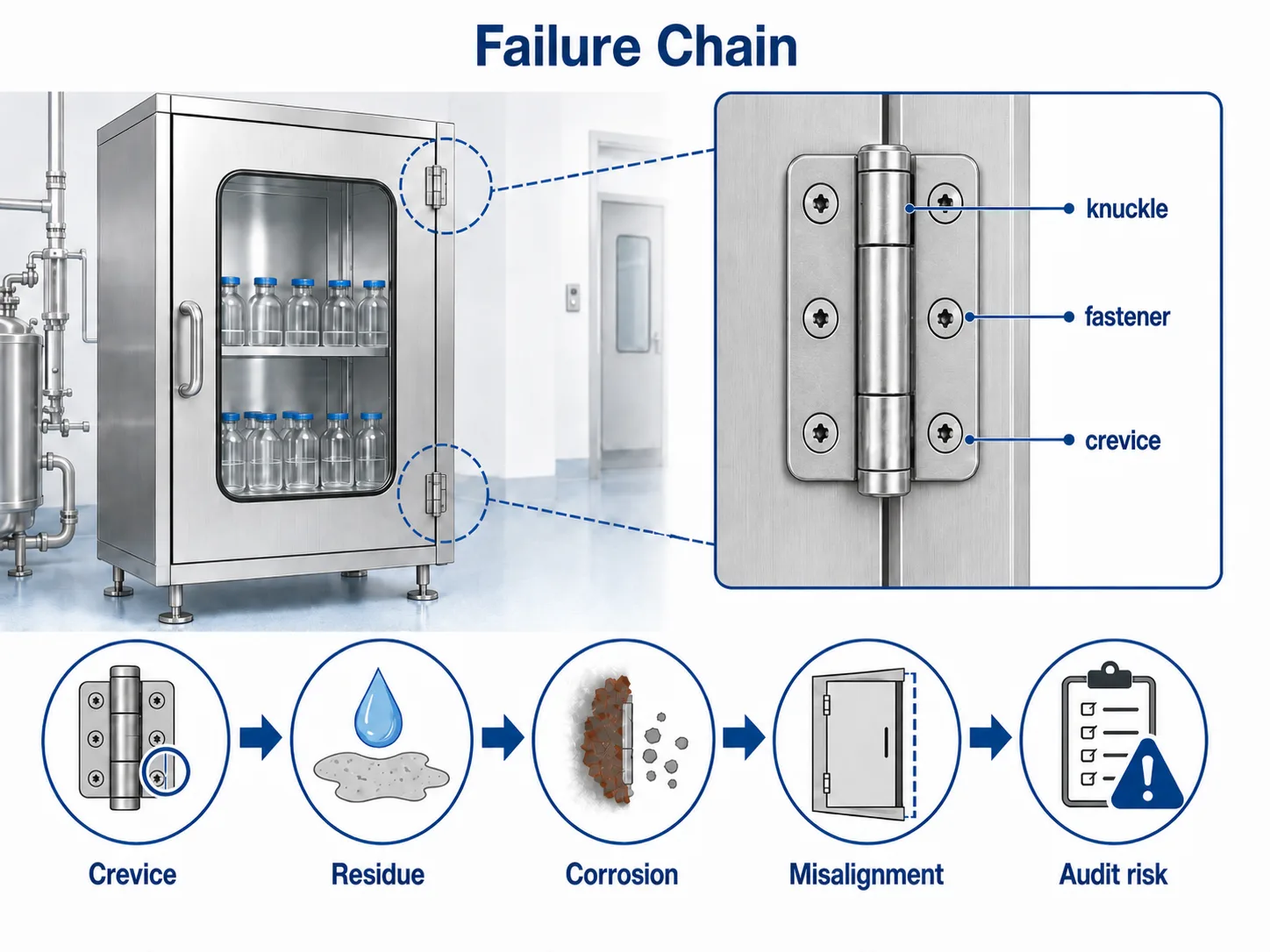
For project teams, the selection question should be:
- Will this hinge be easy to wipe and inspect?
- Will the hinge material resist the actual disinfectants used on site?
- Will the hinge structure create hidden gaps or residue traps?
- Will the door remain aligned after repeated access?
- Can the cabinet be cleaned, inspected, or serviced without unnecessary downtime?
If the answer is unclear, the hinge should not be approved only because it fits the mounting holes.
Quick Cleanability Test — A 5-Minute Field Check
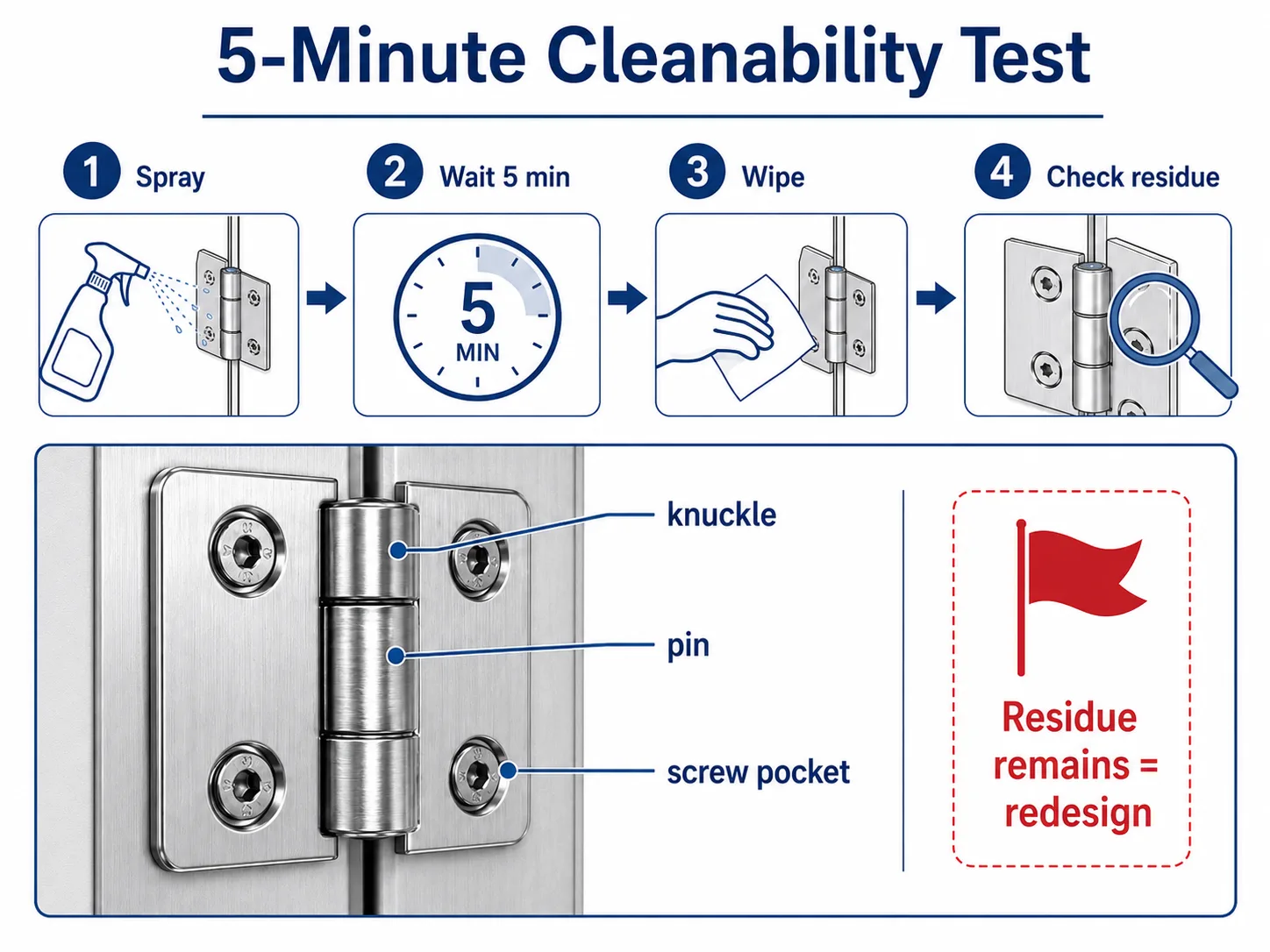
Before approving a hinge sample, run a simple cleanability check: apply the site’s actual disinfectant to the closed hinge knuckle, wait 5 minutes, then wipe the area with a clean lint-free wipe and inspect both the wipe and the hinge. If the wipe shows residue or the knuckle still feels wet around the pin, the hinge geometry is collecting cleaning agent — that is a redesign flag, not a cleaning-procedure problem.
This test takes five minutes, requires no equipment beyond what the cleaning team already uses, and catches the cleanability problems that a paper datasheet review will miss.
Key Selection Criteria for Pharmaceutical Cabinet Hinges
Pharmaceutical cabinet hinge selection should be treated as a hardware specification decision, not an afterthought. The following criteria help buyers and engineers avoid common problems before samples are approved.
Cleanability and Crevice Control
Cleanability is usually the first selection factor. A hinge with exposed pockets, rough edges, sharp corners, open gaps, or difficult-to-reach screw heads can increase cleaning time and make inspection less reliable. Smooth hinge leaves, accessible surfaces, controlled gaps, and simplified mounting details are preferred when cabinets must be wiped down frequently.
Concealed or semi-concealed hinge structures can help reduce exposed hardware on the cabinet exterior, but they must still allow service access and proper door alignment. Continuous hinges can distribute load well on long doors, but their knuckle area should be evaluated for residue retention and cleaning access. The best structure depends on the cabinet’s cleaning procedure, door size, and maintenance needs.
Stainless Grade and Surface Finish
Stainless steel is commonly used for pharmaceutical cabinet hinges because it supports corrosion resistance and cleanable surfaces. SS304 may be suitable for general controlled environments with mild cleaning exposure. SS316 or SS316L is usually safer when the cabinet is exposed to more aggressive disinfectants, chloride residue, washdown, or frequent chemical cleaning.
Material grade alone is not enough. Surface finish, passivation, polishing quality, fastener compatibility, and moisture retention around the hinge also matter. When disinfectants, chloride residue, or trapped moisture collect around hinge pins and screw holes, buyers should understand por qué las bisagras de acero inoxidable aún pueden corroerse before approving 304, 316, or 316L hardware for pharmaceutical cabinets.
Door Load, Alignment, and Access Frequency
Cleanability is important, but the hinge must also support the door mechanically. A pharmaceutical cabinet door that sags, binds, or loses alignment can create latch problems, uneven sealing, repeated adjustments, and operator frustration. Door weight, door width, hinge spacing, gasket compression, and opening frequency should be reviewed before final hinge selection.
For larger pharmaceutical equipment doors, access panels, or stainless cabinet doors with added gasket compression, hinge selection should follow the same system-level logic used for industrial enclosure hinge selection: the hinge, latch, gasket, frame, and maintenance access all need to work together.
Maintenance Access and Removable Doors
Some pharmaceutical cabinets need periodic deep cleaning, filter access, instrument removal, calibration, or maintenance inspection. In these cases, removable-pin, lift-off, or tool-assisted removable hinge designs may reduce service time. However, removable access must not compromise door alignment, cleaning control, or operator safety.
If cabinet doors must be removed during maintenance or deep cleaning, the hinge design should provide controlled removal without loose parts, excessive force, or repeated realignment. For applications where maintenance teams need fast access without damaging the door or gasket, lift-off hinge selection should be evaluated together with cabinet clearance and cleaning procedure.
Application Matrix for Pharmaceutical Equipment Cabinets
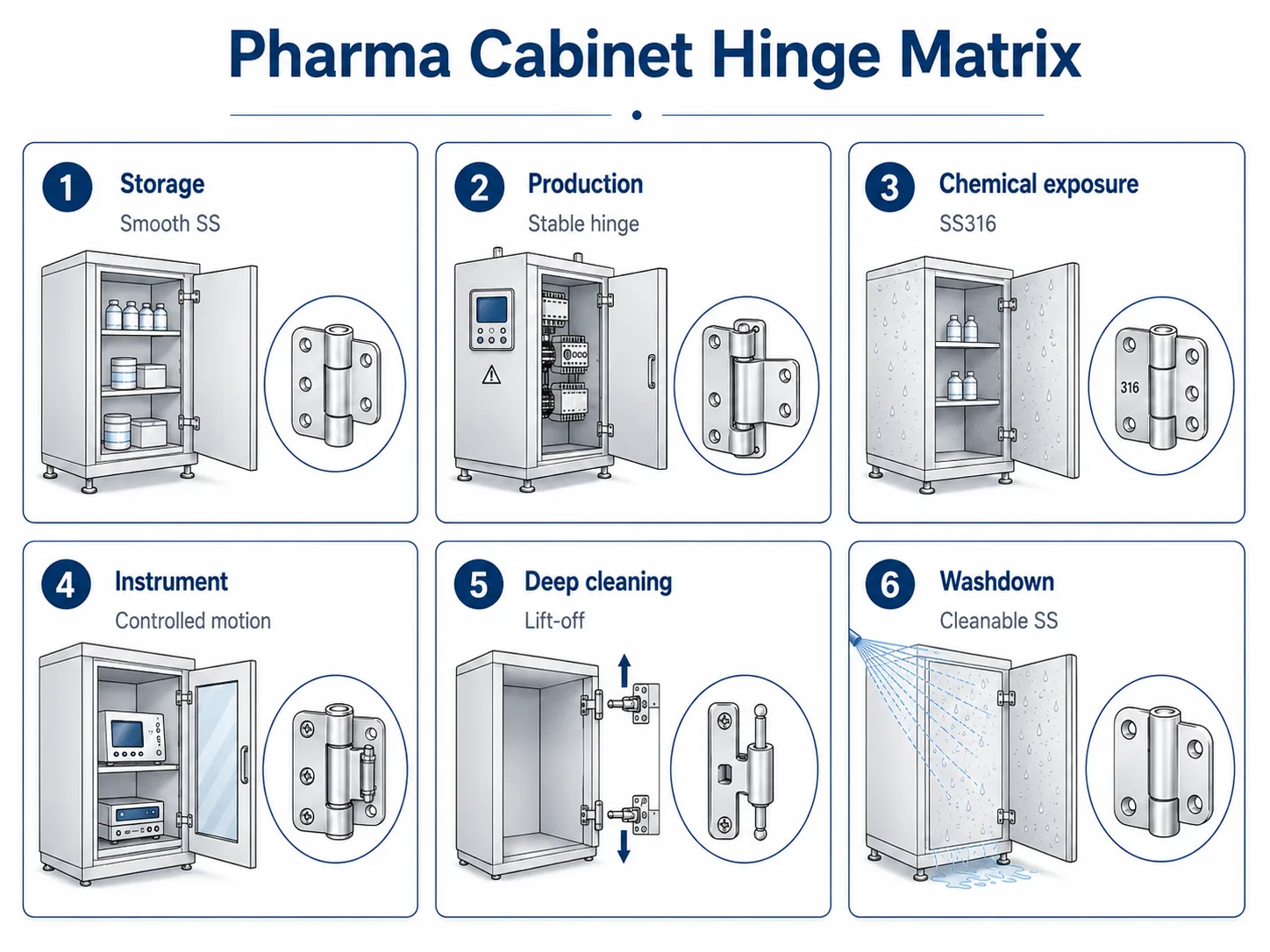
The table below gives a practical starting point. Final hinge selection should still be confirmed against the cabinet drawing, cleaning SOP, door load, room requirements, and supplier documentation.
| Cabinet Application | Riesgo principal | Dirección recomendada de la bisagra | What to Verify |
|---|---|---|---|
| Cleanroom storage cabinet | Residue traps and cleaning difficulty | Stainless hinge with smooth exposed surfaces | Surface finish, screw exposure, cleanability |
| Pharmaceutical production equipment cabinet | Frequent access and door misalignment | SS304 or SS316 hinge with stable alignment | Door weight, hinge spacing, cycle expectation |
| High-frequency access panel | Wear, particles, and pin friction | Serviceable stainless hinge with controlled pin design | Pin wear, lubrication policy, inspection access |
| Disinfectant-exposed cabinet | Pitting, staining, or crevice corrosion | SS316 or SS316L where chemical exposure is aggressive | Cleaning-agent compatibility, passivation, fasteners |
| Instrument or calibration cabinet | Door slamming and vibration transfer | Controlled-motion or damped hinge where needed | Opening force, door control, instrument sensitivity |
| Medical device assembly cabinet | Door positioning and operator ergonomics | Position-control or torque hinge for sustained angles | Holding torque, repeatability, operator access |
| Deep-cleaning access cabinet | Long service time during maintenance | Removable-pin or lift-off hinge if procedure allows | Removal clearance, reinstallation alignment, loose parts control |
| Washdown-adjacent pharmaceutical equipment | Moisture retention and hygiene control | Stainless hinge with cleanable geometry | Drainage, exposed gaps, fastener compatibility |
For cabinets that house medical devices during sub-assembly, calibration, or quality inspection, the door must often hold at a defined open angle for operator workflow. In those cases, selección de bisagras de torsión para dispositivos médicos covers the position-control trade-offs that go beyond cleanability alone.
Pharmaceutical cleaning and food equipment washdown can look similar from a hardware perspective, but the compliance context is different. If the cabinet is part of a food or beverage processing line, selección de bisagras para equipos de procesado de alimentos should be reviewed separately because hygiene, cleaning residue, lubricant control, and equipment access are often evaluated through a different operating lens.
Cleaning-Agent and Corrosion Risk Checklist
Before approving a pharmaceutical cabinet hinge, confirm the cleaning chemicals and wiping frequency used at the site. Stainless grade, surface finish, and fastener material should be selected around the actual cleaning routine, not only the cabinet material.
| Checklist Item | Por qué es importante | Supplier Confirmation Needed |
|---|---|---|
| Cleaning agent type | Some disinfectants are more aggressive toward stainless surfaces or fasteners | Material compatibility statement or application review |
| Wiping frequency | Daily or shift-based cleaning increases exposure | Recommended grade and finish for cleaning frequency |
| Acabado superficial | Rough or damaged surfaces can hold residue | Finish description, polishing/passivation details if required |
| Fastener material | Mixed metals can increase staining or galvanic risk | Fastener grade and compatibility with hinge body |
| Crevice locations | Hidden gaps can trap moisture and cleaning residue | Drawing or sample review of hinge-to-cabinet interface |
| Post-installation inspection | Installation damage can expose edges or create gaps | Installation and inspection checklist |
The goal is not to choose the most expensive stainless grade automatically. The goal is to choose a hinge system that can survive the actual cleaning environment while remaining easy to inspect and maintain.
Common Mistakes When Selecting Pharmaceutical Cabinet Hinges
Mistake 1: Choosing Only by Stainless Grade
Specifying “stainless steel hinge” is not enough. The hinge grade, fasteners, surface finish, exposed gaps, and cleaning-agent compatibility all matter. A poor hinge design in SS316 can still be difficult to clean or inspect.
Mistake 2: Ignoring Screw Heads and Mounting Pockets
Exposed screw heads, recessed pockets, and uneven mounting surfaces can become residue traps. In controlled environments, the mounting interface should be reviewed as carefully as the hinge body.
Mistake 3: Using General Industrial Hinges Without Cleanability Review
A hinge that is strong enough mechanically may still be unsuitable for pharmaceutical equipment if it creates cleaning difficulty, hidden corrosion, or particle risk. Mechanical fit and cleanability must both be evaluated.
Mistake 4: Overlooking Door Alignment After Repeated Access
Frequent access can gradually affect hinge wear and door alignment. If the cabinet door no longer closes consistently, cleaning, sealing, and operator workflow may all be affected.
Mistake 5: Forgetting Maintenance and Replacement Access
Some pharmaceutical cabinets require periodic inspection, calibration, or deep cleaning. If the hinge cannot be inspected, cleaned, adjusted, or replaced without excessive downtime, the selected hinge may increase long-term maintenance cost.
Inspection and Maintenance Schedule
The hinge inspection schedule should match the cabinet’s risk level and cleaning frequency. The table below can be used as a starting point for maintenance planning.
| Frecuencia | Qué comprobar | Por qué es importante |
|---|---|---|
| During routine cleaning | Visible residue, staining, corrosion marks, screw exposure | Detect cleanability problems before they become recurring deviations |
| Monthly | Door alignment, latch engagement, hinge-side gaps | Catch sag or binding before the cabinet becomes difficult to close |
| Quarterly | Pin wear, fastener tightness, surface damage, cleaning access | Identify wear and maintenance needs before downtime occurs |
| After chemical change | Material compatibility, surface reaction, discoloration | Confirm the hinge still matches the updated cleaning procedure |
| Before sample approval or production release | Drawing, material grade, surface finish, mounting interface, cleaning procedure | Prevent unsuitable hinge designs from entering regular production |
Qué enviar a un proveedor de bisagras antes de la selección
To recommend the right hinge for a pharmaceutical equipment cabinet, a supplier needs more than the cabinet size. Provide the cleaning and operating conditions so the hinge can be reviewed as part of the full cabinet system.
- Cabinet application and room environment
- Door size, door weight, and opening frequency
- Cabinet material and surface finish
- Required stainless grade, if already specified
- Cleaning agents and cleaning frequency
- Need for concealed, surface-mounted, continuous, removable-pin, or lift-off structure
- Fastener material and mounting method
- Any gasket, latch, or sealing requirement affected by door alignment
- Inspection or maintenance access requirements
- Sample approval, documentation, or material certificate requirements
For pharmaceutical projects, the best hinge choice is not always the most complex design. It is the hinge that supports cleaning, corrosion resistance, access, alignment, documentation, and long-term service with the least unnecessary risk.
Recomendación final
For pharmaceutical equipment cabinets, start with the cleaning method and cabinet environment, then select the hinge material, structure, and finish. SS304 may be enough for lower-risk controlled areas. SS316 or SS316L should be considered when disinfectant exposure, chloride residue, washdown, or corrosion risk is higher. Concealed or smooth-surface hinge designs may improve cleanability, while continuous hinges may be more appropriate for long or heavy doors.
The safest approach is to treat the hinge as part of the cabinet system. The hinge must support the door mechanically, avoid unnecessary residue traps, resist the cleaning environment, and remain easy to inspect over time. If any of those conditions are uncertain, request a sample review — and run the 5-minute cleanability test described earlier — before approving the hinge for production.
PREGUNTAS FRECUENTES
The best hinge depends on cleaning frequency, disinfectant exposure, door load, cabinet material, and inspection requirements. Stainless steel hinges with smooth surfaces, compatible fasteners, and controlled crevice design are usually preferred for pharmaceutical equipment cabinets.
SS304 may be suitable for general controlled environments with mild cleaning exposure. SS316 or SS316L is usually safer when cabinets are exposed to aggressive disinfectants, chloride residue, washdown, or higher corrosion risk.
Concealed hinges can reduce exposed hardware and improve exterior cleanability, but they are not automatically better for every cabinet. The hinge still needs to support door load, allow service access, and avoid hidden residue traps.
Yes, removable-pin or lift-off hinges can be useful when cabinet doors need deep cleaning, calibration access, or maintenance removal. The design should control loose parts, preserve alignment, and allow safe reinstallation.
Buyers should check stainless grade, surface finish, fastener compatibility, hinge structure, cleaning-agent exposure, door load, mounting interface, inspection access, and supplier documentation before approving pharmaceutical cabinet hinges.
Need Help Selecting Pharmaceutical Cabinet Hinges?
If your project involves pharmaceutical equipment cabinets, cleanroom storage cabinets, controlled laboratory furniture, or stainless equipment doors, HTAN can help review the cabinet environment before hinge selection. Share your cabinet material, door size, cleaning agent, cleaning frequency, required stainless grade, and maintenance access needs, and our engineering team can help recommend a hinge direction that supports cleanability, corrosion resistance, alignment, and long-term service.